
However, he still lacked experimental data on what these particles actually were, and hence undertook a third experiment to determine their basic properties. From these two experiments, Thomson concluded, "I can see no escape from the conclusion that (cathode rays) are charges of negative electricity carried by particles of matter." Under these circumstances, the cathode rays did bend with the application of an electric field. Thomson theorized that the traces of gas remaining in the tube were being turned into an electrical conductor by the cathode rays themselves, and managed to extract nearly all of the gas from the tube to test his hypothesis. A charged particle will curve as it moves through an electric field, but not if it is surrounded by a conducting material. Failing this, he concluded that the negative charge and the cathode rays were somehow stuck together.Īll previous attempts to bend cathode rays with an electric field had failed, so Thomson devised a new approach in a second pivotal experiment. The purpose was to determine if, by bending the rays with a magnet, Thomson could separate the charge from the rays. First, in a variation of a pivotal 1895 experiment by Jean Perrin, he built a pair of cathode ray tubes ending in a pair of metal cylinders with a slit in them, which were in turn connected to an electrometer. Three of his experiments proved especially conclusive. Thomson refined previous experiments and designed new ones in his quest to uncover the true nature of these mysterious cathode rays. Scientists theorized that the glow was produced by some kind of ray emitted by the cathode, but it took the seminal research of a British professor in Cambridge University's Cavendish Laboratory to finally provide a solution to the puzzle. The result: the interior of the tube would glow in lovely fluorescent patterns. They took a glass tube with wires embedded in opposite ends, administered a high voltage and pumped out most of the air. 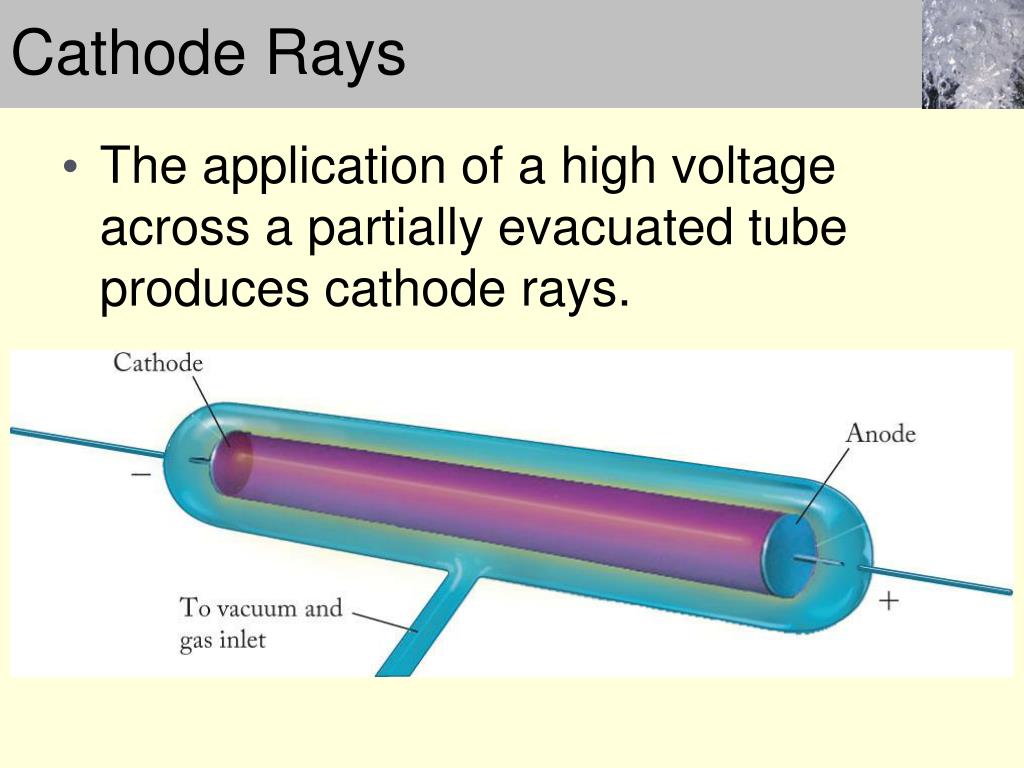
Science lecturers traveling from town to town in the mid-19th century delighted audiences with a device that could be considered the ancestor of the neon sign. As a result of his gold foil experiment, Rutherford’s atomic theory holds good even today.October 1897: The Discovery of the Electron In this model, a vastly empty atom holds a tiny nucleus at the center surrounded by a cloud of electrons. He further went on to reject the plum pudding model and developed a new atomic structure called the planetary model. He also used materials other than gold for the foil, though the gold foil version gained the most popularity. Rutherford named it the “nucleus” after experimenting with various gases. In addition to the above, Rutherford concluded that since the central core could deflect the dense alpha particles, it shows that almost the entire mass of the atom is concentrated there. Most of the particles moved undeviated as there was no obstruction to their path, proving that the majority of an atom is empty. The particles that got deflected by huge angles passed close to the said concentrated mass. The unexpected outcome could have only one explanation – a highly concentrated positive charge at the center of an atom that caused an electrostatic repulsion of the particles strong enough to bounce them back to their source. Simulation of Rutherford’s Gold Foil ExperimentĬourtesy: University of Colorado Boulder Conclusion There were about 1 in every 2000 particles that got scattered by a full 180 degree, i.e., they retraced their path after hitting the gold foil. Though most of the alpha particles behaved as expected, there was a noticeable fraction of particles that got scattered by angles greater than 90 degrees. Like the plum pudding model, since the positive charge of atoms was evenly distributed and too small as compared to that of the alpha particles, the deflection of the particulate matter was predicted to be less than a small fraction of a degree. Since the positively charged alpha particles possess mass and move very fast, it was hypothesized that they would penetrate the thin gold foil and land themselves on the screen, producing fluorescence in the part they struck. A circular screen coated with zinc sulfide surrounded the foil. They used gold since it is highly malleable, producing sheets that can be only a few atoms thick, thereby ensuring smooth passage of the alpha particles. Alpha particles are positively charged particles with a mass of about four times that of a hydrogen atom and are found in radioactive natural substances. They bombarded a thin gold foil of thickness approximately 8.6 x 10 -6 cm with a beam of alpha particles in a vacuum. The method used by scientists included the following experimental steps and procedure.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
